What can waning efficacy look or sound like?
Over time, repeated exposure to neurotoxins with complexing proteins can contribute to a decrease in treatment response.
Waning Efficacy and Neutralizing Antibodies
The FDA has issued guidance for the industry to conduct immunogenicity* evaluations of their products to appropriately assess the risk of NAb2:

Development of antibodies can limit product efficacy in patients treated with therapeutic protein products. NAb can block the efficacy of therapeutic protein products by specifically targeting domains critical for efficacy.”2
- FDA Guidance on Immunogenicity Assessment for Therapeutic Protein Products
Signs of Waning Efficacy with Chronic, Long-term Exposure to Complexing Proteins in Neurotoxin Formulations
NAb formation does not occur in all patients, but because of its increasing likelihood with long-term neurotoxin use, it should be considered when starting or adjusting therapy.1
COMPLEXING PROTEINS MAY CONTRIBUTE TO NAb FORMATION


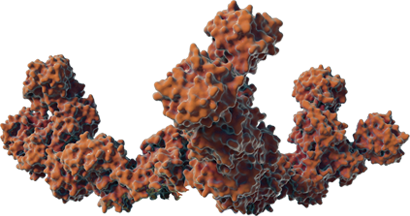

The FDA defines immunogenicity as “the propensity of a therapeutic protein product to generate immune responses to itself and to related proteins or to induce immunologically related adverse clinical events.”2
The direct impact of the non-therapeutic proteins on long-term safety or efficacy continues to be evaluated. Information about the unique XEOMIN manufacturing process and the properties of incobotulinumtoxinA is not intended to imply superiority over other botulinum toxin type A products.
Neutralization is possible with all neurotoxins with or without complexing proteins.
FDA, US Food and Drug Administration; NAb, neutralizing antibodies.